
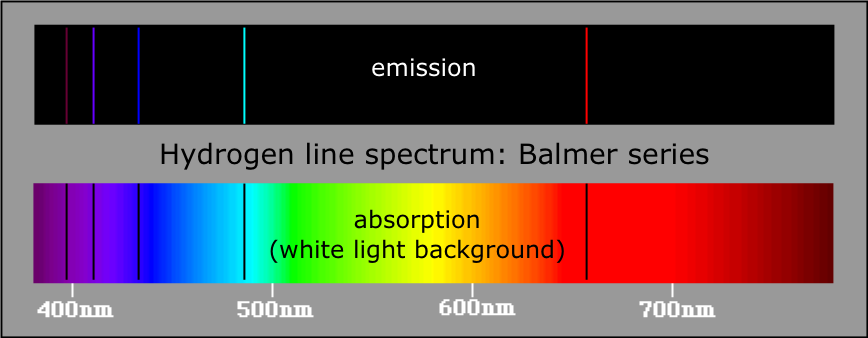
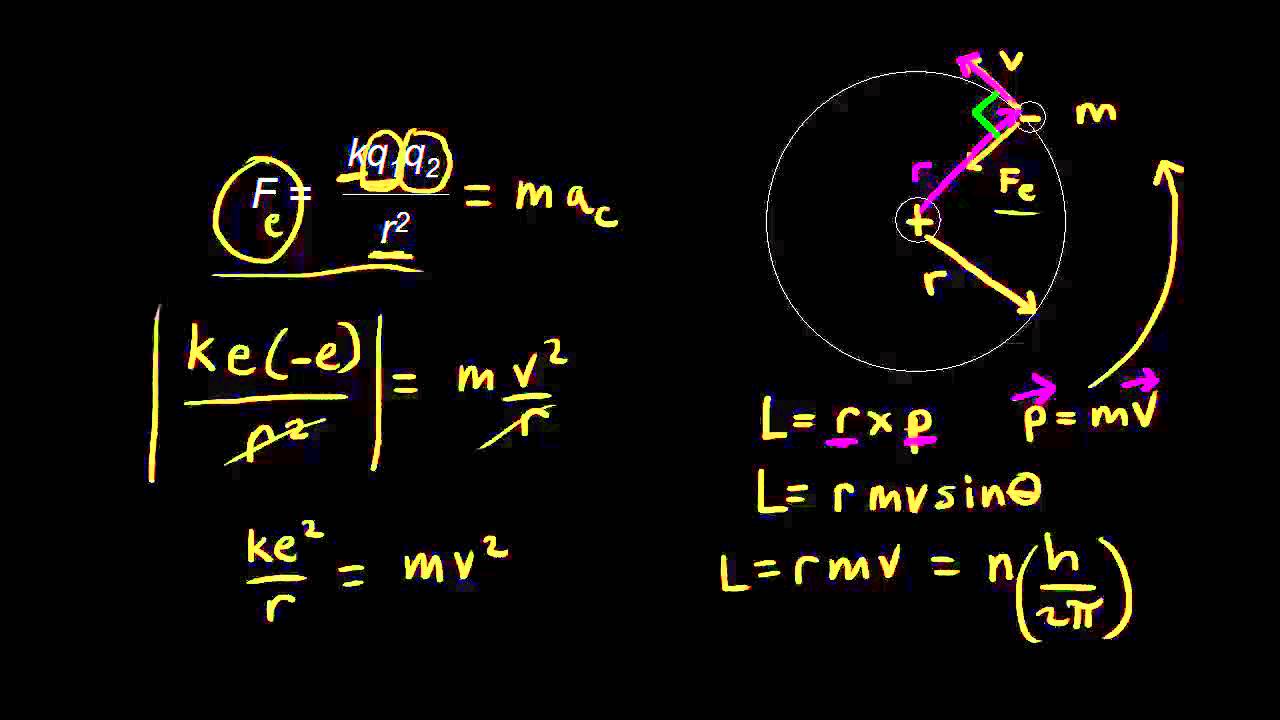
On the basis of Bohr's theory, derive an expression for the radius of the of the n^th orbit of an electron of hydrogen atom.
Atomic spectra, simple models of atoms | Introduction to the physics of atoms, molecules and photons

On the basis of Bohr's theory, derive an expression for the radius of the of the n^th orbit of an electron of hydrogen atom.

If in Bohr's model, for unielectronic atoms, the following symbols are used, rn,z→ Radius of n^th orbit with atomic number Z ; Un,z→ Potential energy of e^- ; Kn,z→ Kinetic energy of

In Bohr's model, for aunielectronic atom, following symbols are used r(n)z rarr Potential energy n+(th) orbit with atomic number Z, U(n,z) rarr Potantial energy of election , K(n,z)rarr Kinetic energy of electron ,

B&I Messezeitung zur IN.STAND 2022, Ausgabe 5 (September) by B&I Die Industrie-Zeitung | Betriebstechnik und Instandhaltung - Issuu













![download as PDF [9.4MB] - Niels Bohr Institutet - Københavns ... download as PDF [9.4MB] - Niels Bohr Institutet - Københavns ...](https://img.yumpu.com/18272665/1/500x640/download-as-pdf-94mb-niels-bohr-institutet-kbenhavns-.jpg)